43 how can proteins be denatured
› protocols › sample-preparation-forWestern blot sample preparation | Abcam *Proteins that are found exclusively or predominantly in a sub-cellular location will be more enriched in a lysate of the sub-cellular fraction compared with whole cell or tissue lysates. This can be useful when trying to obtain a signal for a weakly-expressed protein. Please consult our separate protocols for sub-cellular fractionation. Protein: acid denaturation - IFST
en.wikipedia.org › wiki › Gel_electrophoresisGel electrophoresis - Wikipedia Gel electrophoresis is a method for separation and analysis of biomacromolecules (DNA, RNA, proteins, etc.) and their fragments, based on their size and charge.It is used in clinical chemistry to separate proteins by charge or size (IEF agarose, essentially size independent) and in biochemistry and molecular biology to separate a mixed population of DNA and RNA fragments by length, to estimate ...

How can proteins be denatured
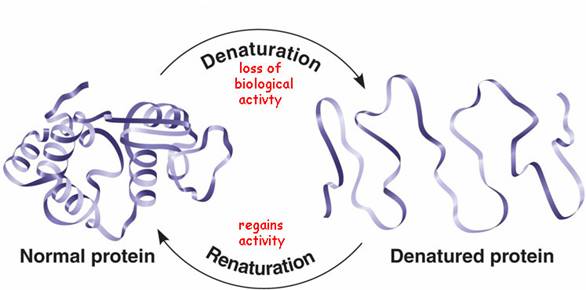
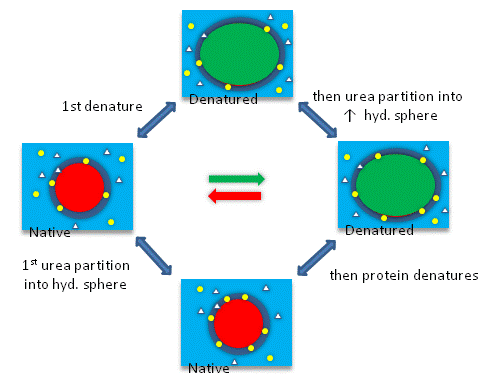
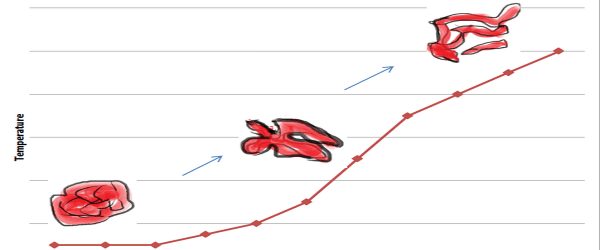
› science › proteinprotein - Protein denaturation | Britannica When a solution of a protein is boiled, the protein frequently becomes insoluble—i.e., it is denatured—and remains insoluble even when the solution is cooled. The denaturation of the proteins of egg white by heat—as when boiling an egg—is an example of irreversible denaturation. The denatured protein has the same primary structure as the original, or native, protein. The weak forces ... en.wikipedia.org › wiki › Denaturation_(biochemistry)Denaturation (biochemistry) - Wikipedia In biochemistry, denaturation is a process in which proteins or nucleic acids lose the quaternary structure, tertiary structure, and secondary structure which is present in their native state, by application of some external stress or compound such as a strong acid or base, a concentrated inorganic salt, an organic solvent (e.g., alcohol or chloroform), agitation and radiation or heat. en.wikipedia.org › wiki › Inverted_repeatInverted repeat - Wikipedia The intervening sequence of nucleotides between the initial sequence and the reverse complement can be any length including zero. For example, 5'---TTACGnnnnnn CGTAA---3' is an inverted repeat sequence. When the intervening length is zero, the composite sequence is a palindromic sequence.
How can proteins be denatured. › articles › nprotUsing circular dichroism spectra to estimate protein ... - Nature Jan 25, 2007 · Circular dichroism (CD) is an excellent tool for rapid determination of the secondary structure and folding properties of proteins that have been obtained using recombinant techniques or purified ... chem.libretexts.org › Courses › University_of2.3: Denaturation of proteins - Chemistry LibreTexts Jul 15, 2019 · Levels of Protein Structure. The structure of proteins is generally described as having four organizational levels. The first of these is the primary structure, which is the number and sequence of amino acids in a protein’s polypeptide chain or chains, beginning with the free amino group and maintained by the peptide bonds connecting each amino acid to the next. en.wikipedia.org › wiki › Inverted_repeatInverted repeat - Wikipedia The intervening sequence of nucleotides between the initial sequence and the reverse complement can be any length including zero. For example, 5'---TTACGnnnnnn CGTAA---3' is an inverted repeat sequence. When the intervening length is zero, the composite sequence is a palindromic sequence. en.wikipedia.org › wiki › Denaturation_(biochemistry)Denaturation (biochemistry) - Wikipedia In biochemistry, denaturation is a process in which proteins or nucleic acids lose the quaternary structure, tertiary structure, and secondary structure which is present in their native state, by application of some external stress or compound such as a strong acid or base, a concentrated inorganic salt, an organic solvent (e.g., alcohol or chloroform), agitation and radiation or heat.
› science › proteinprotein - Protein denaturation | Britannica When a solution of a protein is boiled, the protein frequently becomes insoluble—i.e., it is denatured—and remains insoluble even when the solution is cooled. The denaturation of the proteins of egg white by heat—as when boiling an egg—is an example of irreversible denaturation. The denatured protein has the same primary structure as the original, or native, protein. The weak forces ...
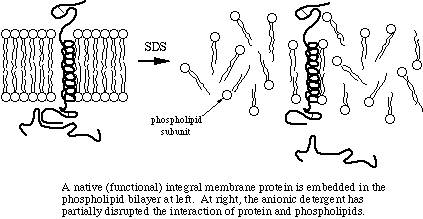
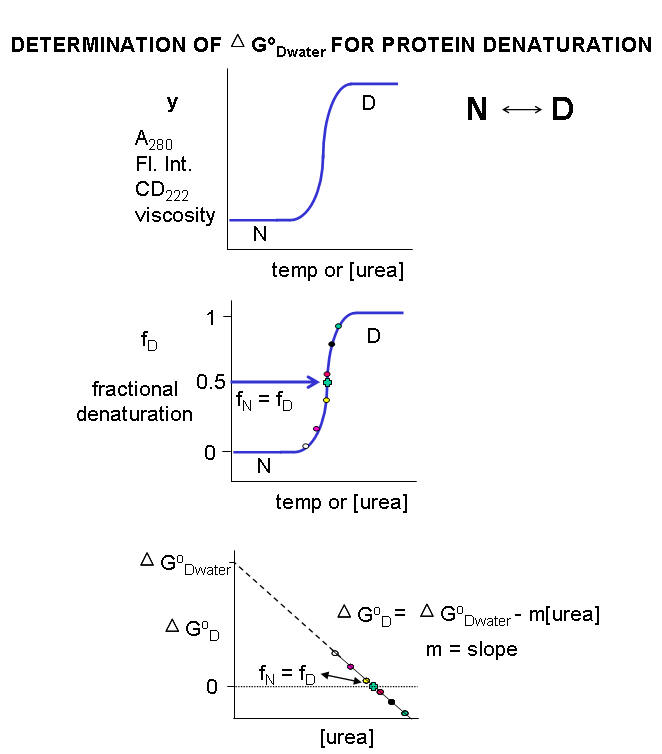












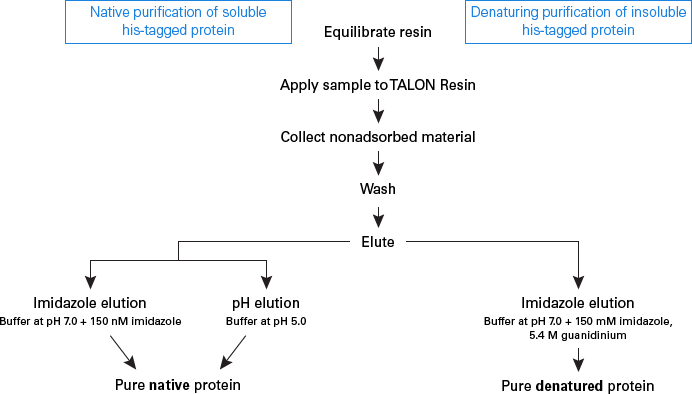

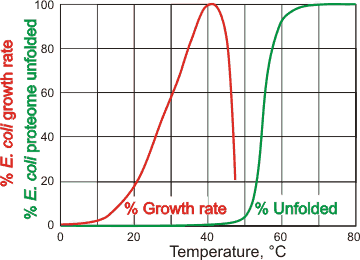

![Protein denaturation [11]. | Download Scientific Diagram](https://www.researchgate.net/publication/320058052/figure/fig2/AS:543270813081600@1506537263689/Protein-denaturation-11.png)










0 Response to "43 how can proteins be denatured"
Post a Comment