42 rate and order of h2o2 decomposition lab answers
General Chemistry 1 PDF | PDF | Gases | Molecules - Scribd (LAB)Determine the effect of various STEM_GC11CK-IIIi-j-139 factors on the rate of a reaction Fourth Quarter General Chemistry 2 Chemical Thermodynamics spontaneous change, prepare a poster on a 1. predict the spontaneity of a process based 1. IB Diploma Chemistry HL Textbook pdf - Academia.edu TITRIMETRIC MTHODS Titrimetric methods are widely used in chemistry to determine oxidants, reductants, acids, bases, metal ions, etc. Titration is based on a reaction between the analyte (unknown sample) and the regent of known concentration and reaction stoichiometry.
An Introduction to Reactive Oxygen Species - BioTek Measurement of lipid peroxidation has historically relied on the detection of thiobarbituric acid (TBA) reactive compounds such as malondialdehyde generated from the decomposition of lipid peroxidation products [25]. While this method is controversial in that it is quite sensitive, but not necessarily specific to MDA, it remains the most widely used means to determine lipid …

Rate and order of h2o2 decomposition lab answers
130+ million publications organized by topic on ResearchGate Explore science topics to find research in your field such as publications, questions, research projects, and methods. Fundamentals of Analytical Chemistry- 9th Edition - Academia.edu Each of the components in a mixture containing a strong acid and a weak acid (or a strong base and a weak base) can be determined provided that the concentrations of the two are of the same order of magnitude and that the dissociation constant for the weak acid or base is somewhat less than about 10 24. chem lab Flashcards | Quizlet Which one of the following sets of units is appropriate for a second-order rate constant? ... You will need to generate a set of graphs to answer this question. Concentration vs time data for the following decomposition reaction was collected at 300 K. The data is shown in the table below. What is the rate constant for the reaction at this temperature (in units of min-1)? H2O2(aq) → …
Rate and order of h2o2 decomposition lab answers. (PDF) monica clinical laboratory part 2 - Academia.edu monica clinical laboratory part 2 How catalyst affect activation energy - batteryfactory.shop In catalytic H 2 –D 2 equilibration, good correlation between the activation energy and the position of the d-band center was observed, experimentally demonstrating the validity of d-band theory. This small amount of energy input necessary for all chemical reactions to occur is called the activation nenergy (or free energy of activation) and is abbreviated E A. Activation energy: … chem lab Flashcards | Quizlet Which one of the following sets of units is appropriate for a second-order rate constant? ... You will need to generate a set of graphs to answer this question. Concentration vs time data for the following decomposition reaction was collected at 300 K. The data is shown in the table below. What is the rate constant for the reaction at this temperature (in units of min-1)? H2O2(aq) → … Fundamentals of Analytical Chemistry- 9th Edition - Academia.edu Each of the components in a mixture containing a strong acid and a weak acid (or a strong base and a weak base) can be determined provided that the concentrations of the two are of the same order of magnitude and that the dissociation constant for the weak acid or base is somewhat less than about 10 24.
130+ million publications organized by topic on ResearchGate Explore science topics to find research in your field such as publications, questions, research projects, and methods.
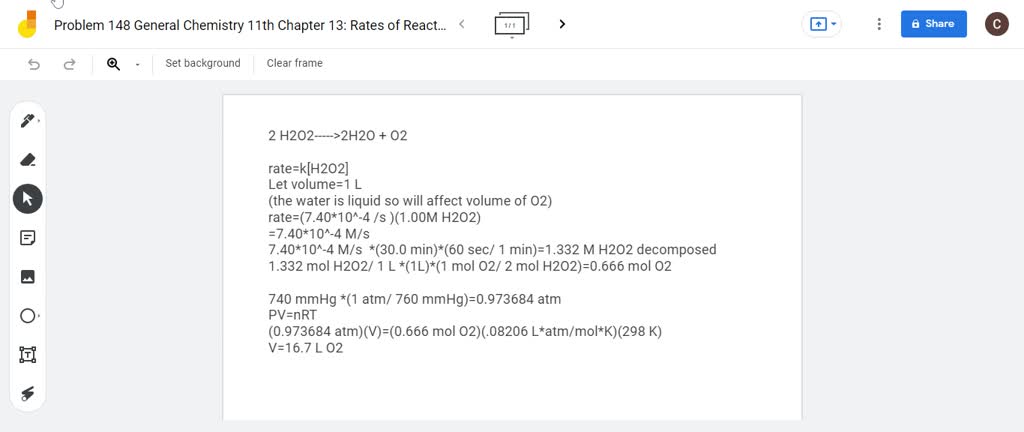
The decomposition of hydrogen peroxide in dilute sodium, hydroxide at 20°C is first order is hydrogen peroxide. H2O2(aq)→, H2O(l) + 1 2 O2(g). During one experiment it was found that when, the initial ...
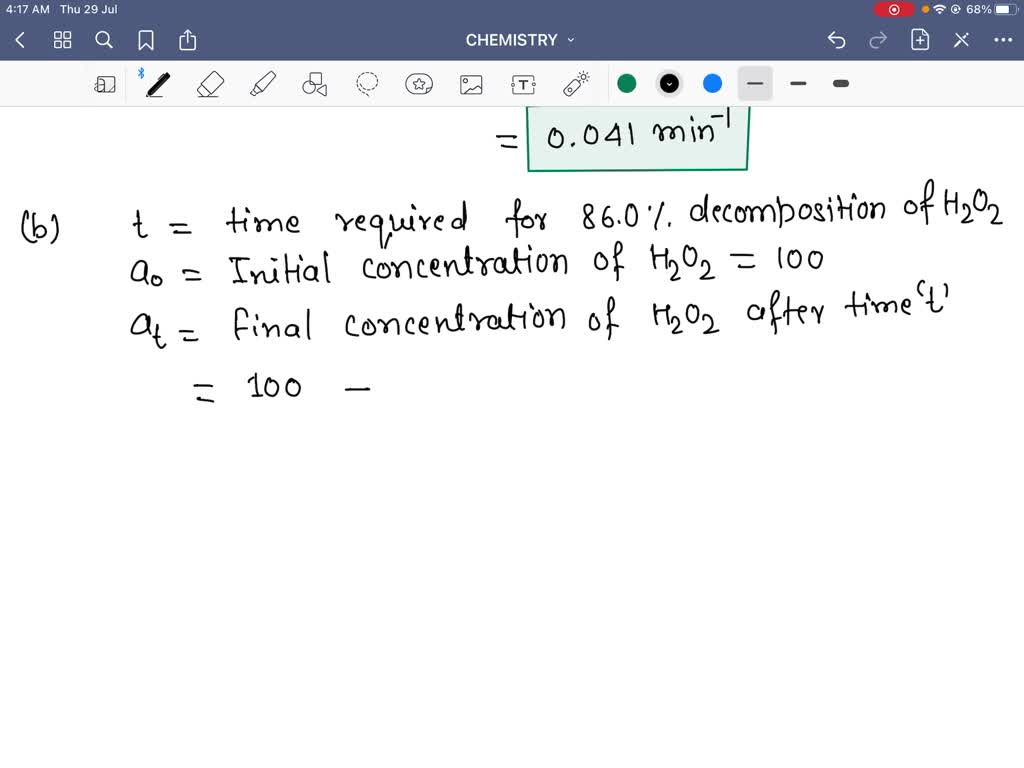
The decomposition of hydrogen peroxide is a firstorder reaction: H2 O2(a q) ⟶H2 O(l)+(1)/(2) O2(g) The half-life of the reaction is 17.0 minutes. a. What is the rate constant of the reaction? b. If ...
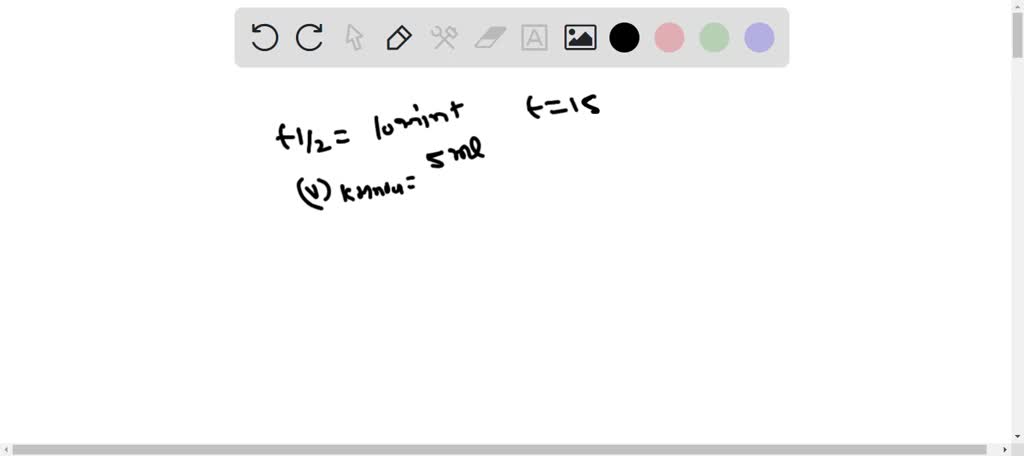
In Part 1 of this experiment, the decomposition of, H2O2 is studied by determining the, concentration of hydrogen peroxide at various times during the, reaction by titration with potassium ...
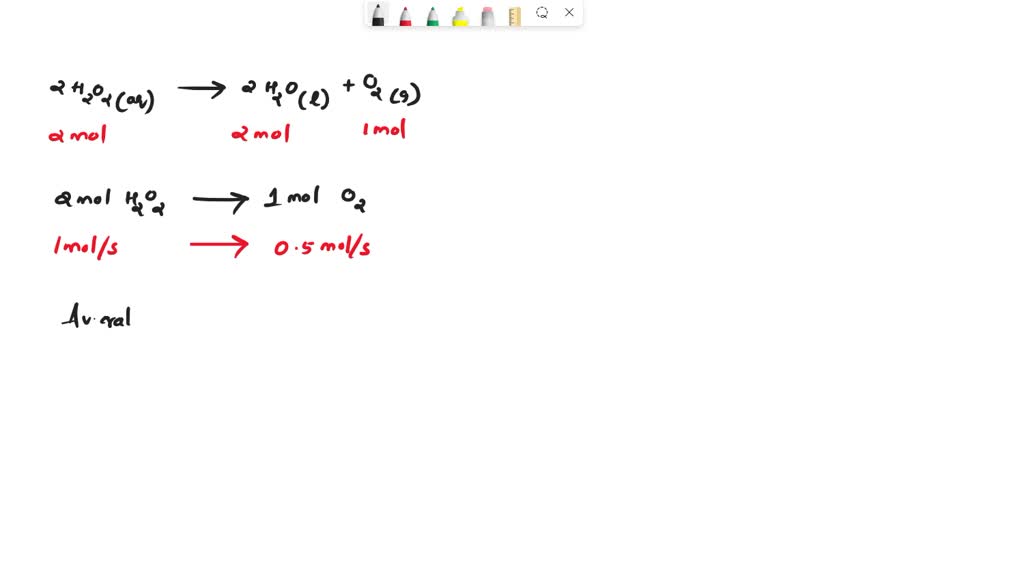
The decomposition of hydrogen peroxide in dilute sodium, hydroxide at 20 °C, H2O2(aq)H2O(l), + ½ O2(g), is first order, in H2O2 with a, rate constant, of 1.10×10-3 min- 1. If an experiment is ...


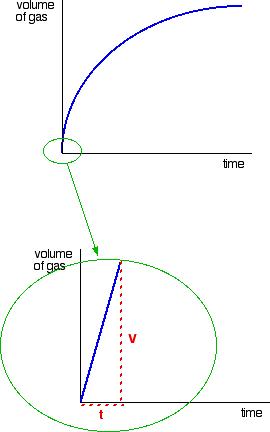




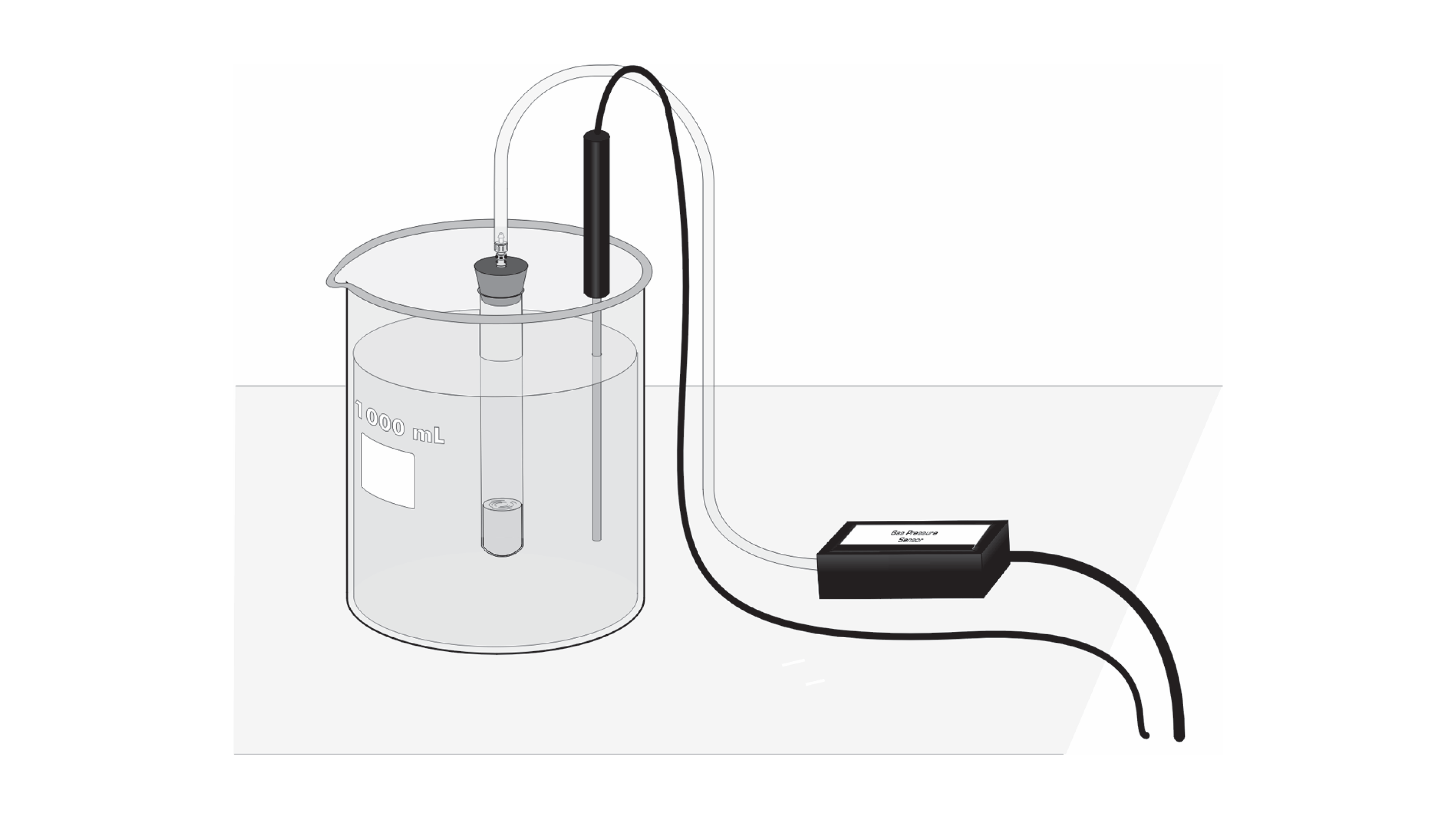






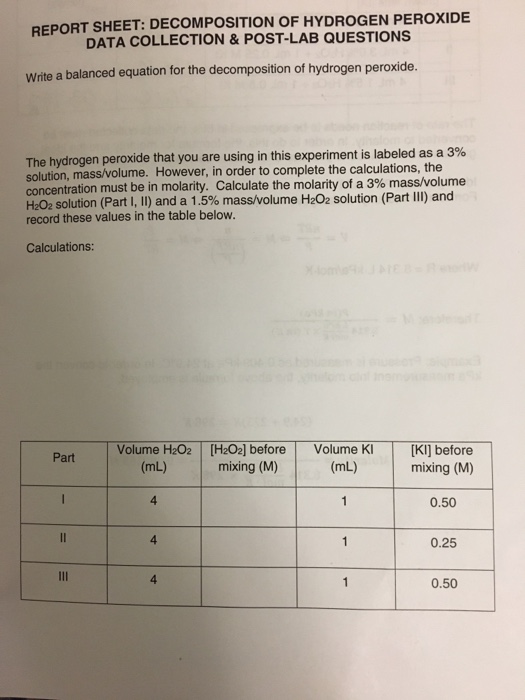











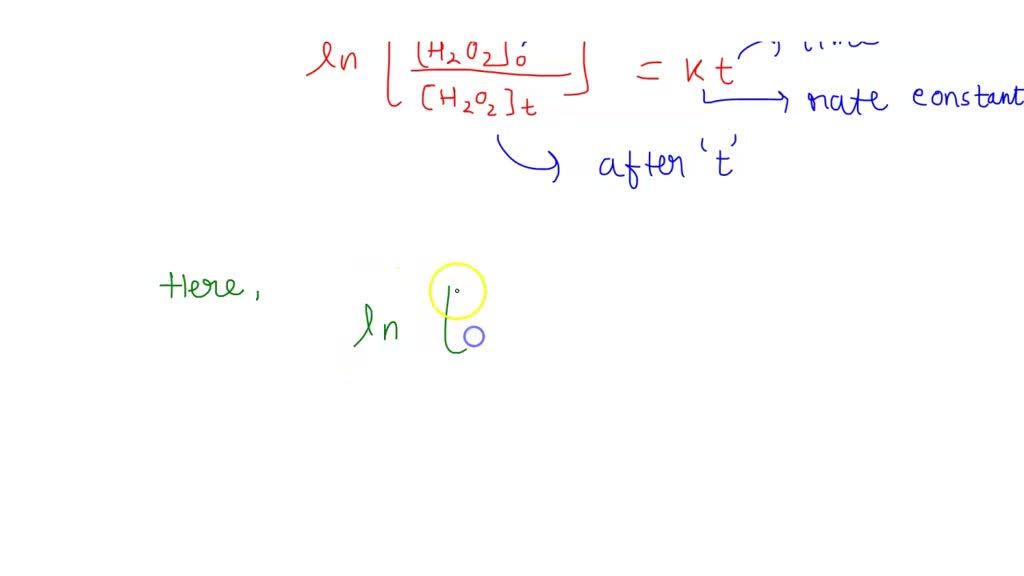
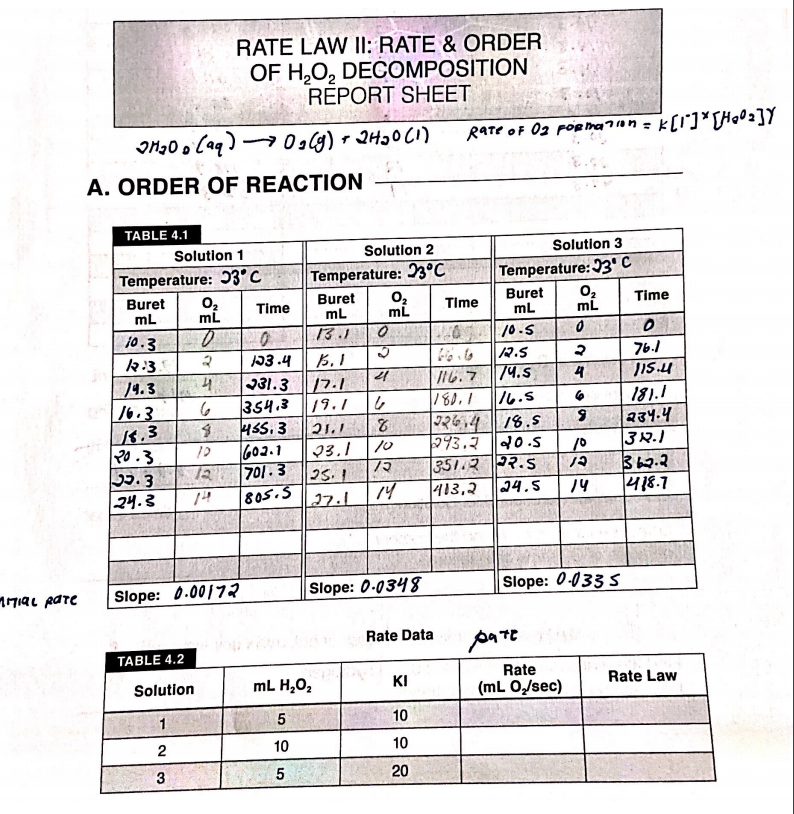

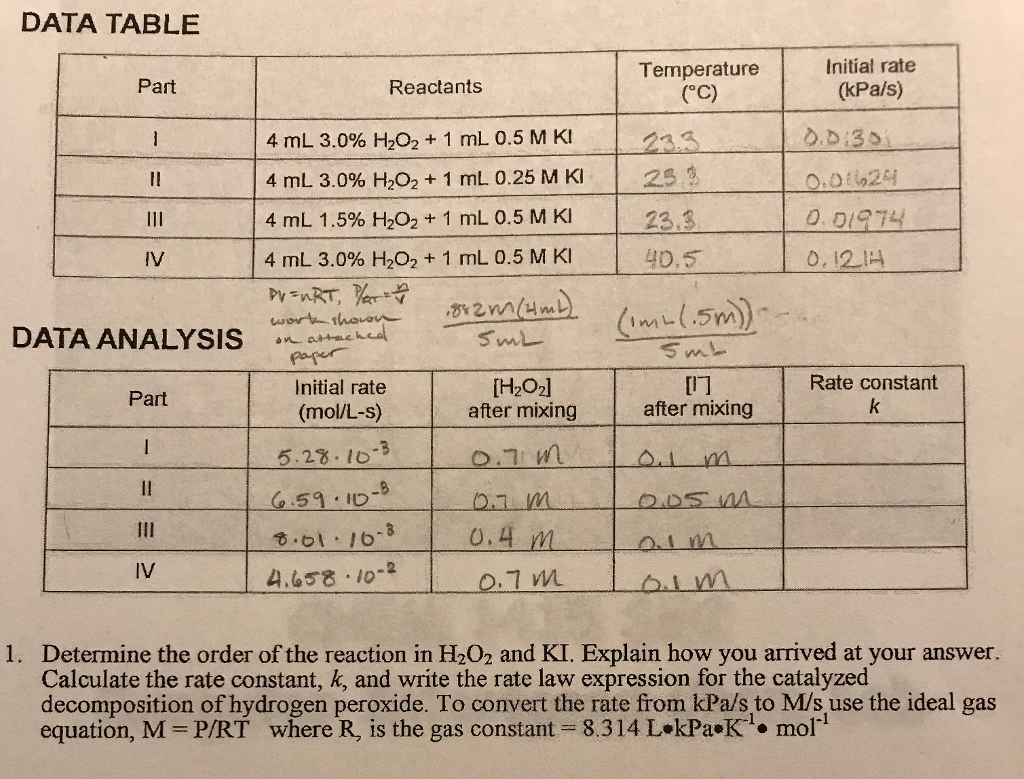

0 Response to "42 rate and order of h2o2 decomposition lab answers"
Post a Comment